The Importance of pH in Cosmetics
pH plays a bigger role in your DIY cosmetics than you might think. Learn how it affects your skin and hair, and how to test and adjust it in your recipes.

If you’ve ever wondered why I don’t recommend soap for hair, or why certain preservatives only work in specific recipes, pH is a big part of the answer.
In this post, I’ll explain what pH is, why it matters in DIY cosmetics, and how to test and adjust it when needed.
Quick summary
- pH measures how acidic or alkaline a water-based product is
- Skin products usually work best around pH 5–6
- Hair products should be slightly more acidic (around 4.5–5.5)
- High pH can damage hair by lifting the cuticle
- Always check the pH range required for your preservative
What is pH?
To understand why pH is important, we first need to understand what it is.
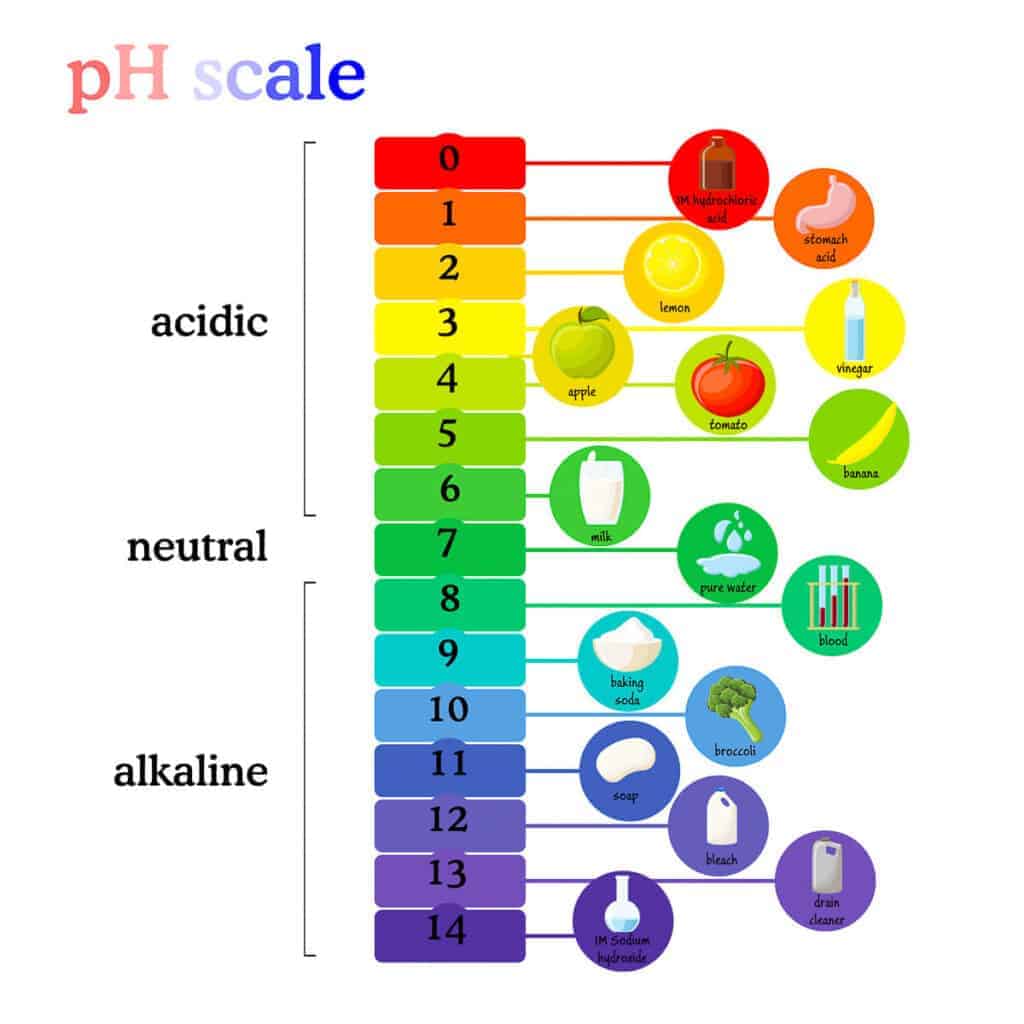
pH is a measure of how acidic or alkaline a water-based solution is, on a scale from 0 to 14. It’s based on the concentration of hydrogen ions in a solution, which is why it can be affected slightly by factors like temperature.
A pH of 7 is considered neutral (like distilled water). Values below 7 are acidic, while values above 7 are alkaline.
In general, the lower the number, the more acidic the solution is, and the higher the number, the more alkaline it is. Strong acids have very low pH values, while strongly alkaline substances have very high ones.
Both extremes can be very caustic and irritating, which is why pH becomes especially important when making products for skin and hair.
pH of skin
While pH is technically a measure of water-based solutions, you’ll often hear people talk about the pH of our skin and say that it’s acidic.
It’s not exactly that the skin itself is acidic, but rather that it’s covered with a mixture of sweat, oil, and beneficial bacteria. These form what’s known as the acid mantle, a slightly acidic layer that helps protect the skin.
When the acid mantle is disrupted or removed, the body is usually able to restore it fairly quickly.
Babies are born with a more neutral skin pH. As they age, the acid mantle develops and their skin becomes more acidic. pH can also vary depending on the area of the body. More exposed areas tend to have a slightly higher pH, while more protected areas (like the armpits or genital area) tend to maintain a lower pH.
Most people are able to recover from temporary changes in skin pH without long-term issues. That’s why using something like soap, which is more alkaline, doesn’t always cause obvious problems.
Some people, though, find those changes more irritating, especially if they have sensitive skin. In those cases, it’s usually best to use cleansers sparingly. When needed, a gentle, non-soap cleanser with a lower pH is often a better option.
Ideal pH for skin products
Generally, a pH of around 5–6 is a good range to aim for when making cleansers and lotions for the skin, as it’s close to the skin’s natural pH.
Staying within this range helps support the skin’s protective acid mantle and reduces the likelihood of irritation, especially for those with sensitive skin.
pH of Hair
While skin can usually recover quickly from changes in pH, hair isn’t as resilient. Apart from the follicle, hair is mostly made up of dead cells and can’t repair itself in the same way skin can.
If you’ve followed my blog for a while, you probably know that I love making soap. Despite that, I don’t use it on my hair. This isn’t just a preference. It’s because of how pH affects the structure of the hair itself.
High pH damages the hair cuticle
Just as skin has a protective layer called the acid mantle, hair has a protective layer called the cuticle. Under a microscope, the cuticle is made up of overlapping scales that lie flat along the hair shaft. When these scales are smooth and in place, they help retain moisture and give hair a softer, shinier appearance.
When you use alkaline products on your hair, those cuticle scales lift and open. This leaves the hair more prone to dryness and breakage. Hair with a raised cuticle is also more likely to tangle and look dull or frizzy.
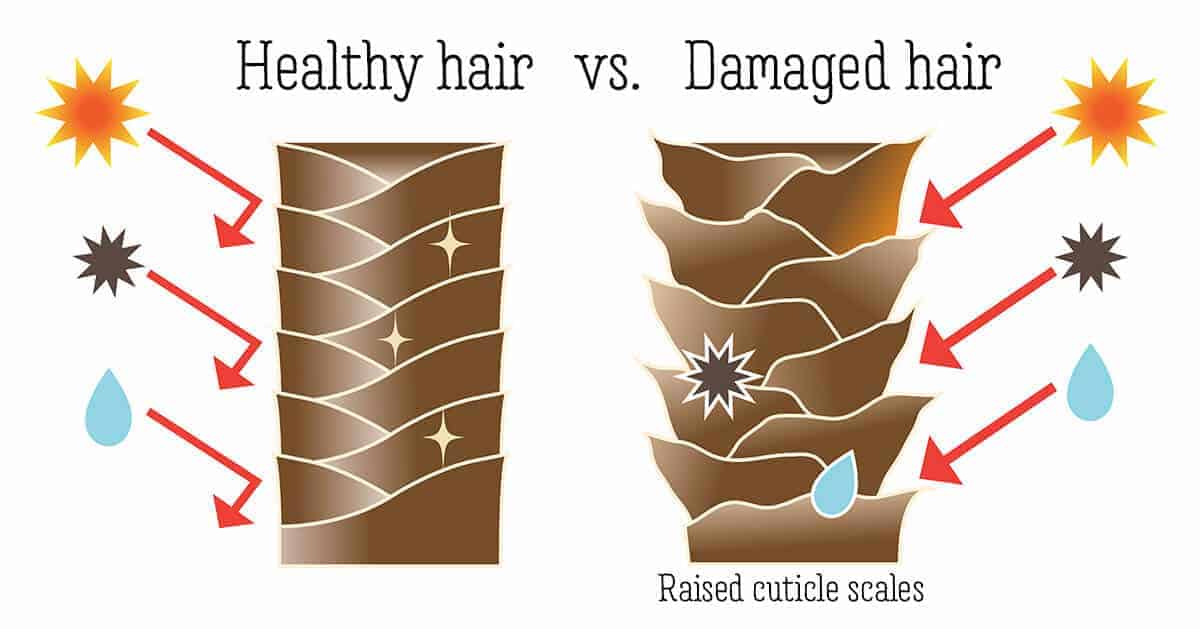
In an attempt to fix that problem, sellers of soap-based “shampoo bars” often recommend a final rinse with vinegar to lower the pH again. This can temporarily improve how the hair looks by helping the cuticle lie flatter, making the hair appear smoother and shinier.
Over time, though, repeatedly raising and lowering the cuticle can leave the hair more damaged. Someone with short hair, or someone who only uses soap occasionally, may not notice the effects right away. With frequent use, especially on longer hair, the damage tends to become more noticeable.
Other ways soap can damage hair
The alkaline pH of soap doesn’t just affect the cuticle. It can also increase the negative charge of the hair, which leads to more friction between strands. That added friction can result in more breakage.
Soap can also leave behind residue on the hair. In hard water, it reacts with minerals like calcium to form deposits that build up over time. This buildup can leave hair looking dull and feeling rough, and it can make tangling and breakage more likely.
Ideal pH for hair products
Most shampoos are formulated within a pH range of about 4.5–5.5, which helps keep the cuticle smooth and the hair in better condition.
Children’s shampoos often have a slightly higher pH. The closer a product is to a neutral pH of 7, the less likely it is to sting the eyes, which is why many are marketed as “tear-free.”
I talk more about that in my baby wash and shampoo recipe post.
pH and cosmetic ingredients
The pH of a cosmetic doesn’t just affect how it interacts with your skin or hair. It also affects how the ingredients in your formula behave.
pH and preservatives
If you’re making a product that contains water, you’ll almost always need a preservative to help prevent the growth of bacteria, mold, and other microbes. While mold is often visible, bacterial growth usually isn’t, and it can still lead to irritation or infection.
Not all preservatives work across all pH levels. In fact, most are only effective within a specific pH range. When choosing a preservative, you’ll need to check the recommended pH range from your supplier and make sure your final product falls within it.
Products with very high or very low pH values are often less prone to microbial growth, which is why they can sometimes be self-preserving. In most cases, though, you’ll be formulating products within a milder, skin-friendly range. That means you’ll still need to use a preservative.
For more information, you can check out my beginner’s guide to natural preservatives.

pH effects on surfactants, emulsifiers, and other ingredients
pH doesn’t just affect how a product feels on your skin or hair. It can also affect how well the ingredients in your formula work.
Some ingredients, like emulsifiers and surfactants, are only stable or effective within certain pH ranges. pH can also affect the thickness of a product, especially when using gums or other thickeners.
For example, in my hand sanitizer gel tutorial, I used TEA (triethanolamine) to raise the pH so the mixture would gel properly.
Some surfactants are especially sensitive to pH. Amphoteric surfactants, for example, can change their charge depending on the pH of the formula. At lower pH levels, they may behave more like cationic ingredients, while at higher pH levels, they can act more like anionic ones. In between, they can exist in a zwitterionic state. (I go into more detail about this in my guide to natural surfactants.)
While most cosmetic ingredients work well within the typical pH range used for skin and hair products, it’s still important to check the recommended pH range for any new ingredient you’re using. This becomes especially important if you plan to adjust the pH of your formula significantly.
How to test pH
There are several ways to test the pH of a product.
pH test strips
The easiest and least expensive way to check pH is to use paper test strips. These strips change color when dipped into a water-based solution, and the color can be compared to a chart to estimate the pH.
There are different types of test strips available. Some cover a wide pH range, while others focus on a narrower range and offer more color variations for slightly better accuracy.
Because it can be difficult to match colors exactly, test strips aren’t ideal when you need a precise reading. You can usually get a good estimate, but not an exact value down to a decimal point.
For most DIY cosmetic recipes, though, that level of accuracy is enough. You usually don’t need to know that your product has a pH of exactly 5.4. It’s generally sufficient to know that it falls within a skin- or hair-friendly range.
Kids often enjoy experimenting with pH strips. They’re a simple way to explore how different substances vary in acidity and alkalinity. My son was especially fascinated by how certain foods can change color depending on their pH. I even used that idea to make color-changing homemade food coloring.

pH meters
If you need more precise measurements, you may want to use a pH meter.
pH meters can give more accurate readings than test strips, but they are more expensive and require some upkeep. To stay accurate, they need to be calibrated regularly using buffer solutions.
They also need to be stored properly. Most probes should be kept slightly moist, and if they dry out completely, they can lose accuracy over time.
For most DIY cosmetic projects, a pH meter isn’t necessary. Test strips are usually accurate enough to make sure your product falls within a safe and effective range.
If you don’t plan to use one often, it’s probably not worth the extra cost or maintenance.
Testing the pH of non-aqueous products
Sometimes you may want to check the pH of a solid product or one that doesn’t contain water, like a shampoo bar or a bar of soap. Since pH can only be measured in water-based solutions, you’ll need to mix the product with water to test it.
For the most consistent results, it’s best to use distilled water. If you’re more interested in how the product behaves during normal use, you can also test it with your tap water.
For example, to test a shampoo bar, you can lather it with water and then use a test strip to measure the pH of the lather.
Distilled water has a neutral pH of around 7. Tap water, on the other hand, contains minerals and other substances that can affect the reading. Testing with tap water can give you a better idea of how the product will behave under your normal conditions.

Adjusting the pH
There are times when you may need to adjust the pH of a product. For liquid products, this is usually straightforward. Work slowly, adding small amounts of a pH adjuster and testing as you go so you don’t overshoot and have to correct it again.
For solid products like shampoo bars or conditioner bars, you’ll need to test the pH after the bars have set. If adjustments are needed, they have to be made in the next batch. That usually involves a bit of trial and error, so it’s important to keep track of how much pH adjuster you add each time. That way, once you get the result you want, you can reproduce it consistently.
Lowering the pH
If the pH of your product is too high, you can lower it by adding an acid. Common options include citric acid and lactic acid.
Lactic acid is usually sold as a concentrated liquid. For small batches, you’ll often only need a drop or two to bring the pH down, so it’s best to add it slowly and test frequently.
Citric acid is typically sold in powder or crystal form. To use it, dissolve it in water to make a solution. I like to make a 10% solution by mixing 1 gram of citric acid with 9 grams of distilled water. Using a standard solution like this makes it easier to measure what you’re adding and reproduce your results later.

Lowering the pH of soap
Some people try to lower the pH of soap, especially when making soap-based “shampoo” bars, by adding acidic ingredients. The problem is that soap relies on its alkalinity to function. If you lower the pH too much, it will start to break down and no longer behave like soap.
Soap can’t be acidic or even truly neutral. I go into more detail about this in my post on “neutralizing” liquid soaps.
Raising the pH
While it’s less common, there are times when you may need to raise the pH of a product.
This can be done using small amounts of sodium hydroxide (NaOH) or potassium hydroxide (KOH). If you make soap, you probably already have these on hand.

Some people try using baking soda, but it’s not very effective for this purpose and usually requires much larger amounts to noticeably raise the pH.
Other ingredients, like TEA (triethanolamine) or arginine, are also used to increase pH in certain formulations. For example, I used TEA to raise the pH when working with carbopol to make a hand sanitizing gel.
In some systems, the choice of pH adjuster really matters. For example, with ingredients like Emulsense HC, a cationic emulsifier often used in conditioners, the manufacturer recommends preserving it with Spectrastat G2-N, which works best within a pH range of about 5–5.5. In that case, they suggest raising the pH with arginine, since more common options like NaOH or baking soda aren’t compatible with that system.
Final Thoughts
pH might seem like a small detail, but it affects more than you’d expect, from how your products feel to how well they actually work.
Once you get used to checking and adjusting it, it becomes a normal part of the process. It also makes it much easier to troubleshoot recipes and get consistent results, especially when working with things like shampoos, conditioners, and preserved products.
This post was originally published on March 11, 2021 and was updated and republished in April 2026.









Thanks so much for this PH 101. Despite using a preservative for a styling hair cream I made, the formula grew mold after 2 weeks. So now the PH issue has my attention! The formula is a xanthan gel and has a wee bit of oil and butter pearls (C10-18 Triglycerides). Below is my formula. Maybe the xanthan has a funky effect on preservatives?
90.00% Distilled water
1.50% Xanthan gum
5.00% Grapeseed oil
2.50% Butter pearls
0.50% Liquid Germal Plus
0.50% Fragrance Oil
Thank you for your blog!!
Hello,
No, the Xanthan gum should not be a problem. I use it a lot in my formulations.
I don’t see an emulsifier in your formula. I’m not sure what butter pearls are, though. (Actually, I just looked it up- and it’s definitely not an emulsifier. It seems like it can thicken emulsions or gel oils, but not emulsify.)
You’re probably having issues because you don’t have anything holding the water and oil phases properly together.
ძალიან დიდი მადლობა . მეტად საჭირო ინფორმაცია იყო.
You’re welcome!