Trying to avoid SLS and other harsh surfactants in your cosmetics? There are many mild, natural surfactants available. Learn about the different types of natural surfactants, with a list of my favorites.

What is a surfactant?
There are many types of surfactants and they are used for many different purposes, but they all share one quality: they help increase the wetting properties of a liquid. Surfactants can be found almost everywhere. You can find them in everything from detergents and shampoos to toothpaste and even conditioners.
Surfactant definition:
(A surfactant,) also called surface-active agent, (is a ) substance such as a detergent that, when added to a liquid, reduces its surface tension, thereby increasing its spreading and wetting properties. (Encyclopaedia Britannica)
Surfactant function
Some surfactants are emulsifiers, others are foaming agents (and some may actually do the opposite of those functions). Some act as detergents, while others act as insecticides or fungicides. Some help with solubilizing (small amounts of oils into water, for example) and others help increase viscosity.
How do surfactants work?
Surfactants affect the surface tension of liquids to increase wetting.
Why would you want to increase wetting?
Normally, when you spray water on a surface like a window, rather than spread evenly over the surface, the water will bead up. That’s because of the surface tension of the water. The molecules of the water come together in a stable configuration and are attracted to each other. When you are trying to clean that window, though, that beading isn’t helping you. You want the water to spread evenly over the surface to better clean it. You also want something that can grab onto the grease and dirt on whatever surface you are trying to clean.
Micelles in surfactants
Surfactants affect the surface tension that is making the water bead up rather than spread out. They have a water-loving head and an fat (oil) loving tail. They come together in structures called micelles.

I already explained a bit about how the micelles in surfactants work in my micellar water recipe, but for those who haven’t read that post, let me give you a quick, simplified explanation. The water-loving heads of the micelles bond with the water while the oil-loving tails on the inside of the micelles bond with the grease and grime. That pulls the grease and grime into the center of the micelles out of contact with the water, making them easier to rinse away.
You’ll also find that hot water helps clean better because the hot water helps melt the fats which makes it easier for them to be brought into the micelles.
Types of surfactants
There are four main types of surfactants, each behaving somewhat differently, and some with completely different functions. The detergent-like surfactants tend to be the anionic, non-ionic and amphoteric surfactants. Some cationic surfactants are used as emulsifiers and are great for hair conditioners. (I use BTMS, a cationic surfactant, in my hair conditioner recipe.)
These are classified based on the charge of the polar head of the surfactant which can have a positive charge (cationic), a negative charge (anionic), or no charge (non-inonic). Amphoteric surfactants have both a cationic and anionic part attached to the same molecule.
- Anionic – Anionic surfactants are the most commonly used surfactants because they tend to provide the best cleaning power and the most foam. You’ve probably heard people talking about one of the most commonly used anionic surfactants, SLS (Sodium lauryl sulfate or Sodium Laureth Sulfate). It can be found in everything from shampoos and shower gels to even toothpaste. I’ve also shown you how to make soap (many types by now!), another anionic surfactant.
Anionic surfactants can be harsher on the skin, which is why they are often combined with other types of milder surfactants. - Nonionic – The second most commonly used surfactants are nonionic surfactants. They don’t ionize in water or aqueous solutions. Nonionic surfactants are gentler when cleaning. Because they don’t carry a charge, they are the most compatible with other types of surfactants. Recently, sugar-based nonionic surfactants have been developed to offer a safer, non-toxic alternative to some of the more harsh surfactants on the market up until now.
- Cationic – Cationic surfactants don’t generally give foaming like the other types of surfactants. They are often used in hair care products (mainly conditioners and anti-static products because they don’t provide the foaming for use in shampoos) because their positive charge is attracted to the negative charge in hair. This makes it difficult to completely wash them from your hair, so some stays behind to help reduce friction between hairs which, in turn, reduces the amount of electrostatic charge in hair. This helps make hair more manageable and helps prevent damage.
Cationic surfactants aren’t usually compatible with anionic surfactants! - Amphoteric – Amphoteric surfactants can carry either a positive or negative charge depending on the pH of your product. Despite that, they are still compatible with all of the other types of surfactants. These tend to be very mild surfactants which is why they are usually combined with other surfactants. While amphoteric surfactants may not give a lot of foam on their own, they can help boost the foam of the other surfactants. Amphoteric surfactants are often combined with anionic surfactants to reduce their harshness and help stabilize their foam.

Natural surfactants list:
Natural surfactants can be derived from many types of plants. Common sources are coconut or palm, but they can also be derived from other types of fruits and vegetables.
There are many natural surfactants on the market today, and with increased consumer demand, I imagine that many more will be available in time. I have tried many of them, but today I’ll focus on some of my favorites. I like these surfactants because they are gentle, they tend to be easier to find, and they work well together. You can use these in everything from gentle shampoos to shower gels, facial cleanser, and baby washes.
Choosing your surfactants
Keep in mind that many of these surfactants are not palm free, so you’ll want to source them from places that allow for sustainable methods of obtaining their materials. I buy surfactants that have been certified sustainable by RSPO (Roundtable on Sustainable Palm Oil) standards.
Another thing to keep in mind is that these surfactants can differ from manufacturer to manufacturer. The names are polymeric and aren’t referring to an exact structure. Some places will use different plants as the origin of elaborating each surfactant, and the way each surfactant cleans, solubilizes, etc. can vary depending on where you buy it from. I’ll be describing these surfactants based on my suppliers, but you’ll want to check on the specifications of the surfactant you are buying if it’s important to you to know what plants have been used to derive them, the pH, the concentration, etc. Use this list as a general guideline!
Along those lines, while mine are listed as ECOCERT approved, that may also be dependent upon the manufacturer of each surfactant.
I’ll be updating this list and adding more surfactants as I use them and learn more about them. For now, though, this should give you a good starting point to understanding what we are going to be working with.
Coco Glucoside
Coco Glucoside is a non-ionic surfactant that is obtained from coconut oil and fruit sugars, but it can also be obtained from either potato or corn. It is a very gentle, foamy cleanser and is completely biodegradable. You can use it in products that you want to have an ECOCERT certification. It has an alkaline pH (around 12) which makes it self-preserving as is, but you will probably have to adjust the final pH of products using it to pull it into a range more suitable for your skin or hair (and you’ll need to add a preservative).
Decyl Glucoside
Decyl Glucoside is very similar to coco glucoside (non-ionic and ECOCERT compatible), but it has a shorter chain length. It creates less foam (its foam is less stable) than coco glucoside but it does add more viscosity to a product. It is derived from coconut oil and glucose and is completely biodegradable. It can be used in all sorts of shampoos, gels, baby products, etc.
Lauryl Glucoside
Lauryl Glucoside is very similar to the other 2 glucosides I’ve mentioned. It has a longer chain length and more viscosity. It takes longer to foam than the other two, but it also has the most stable foam. While it is also a mild cleanser, it isn’t as mild as the other 2 alkyl polyglucosides.
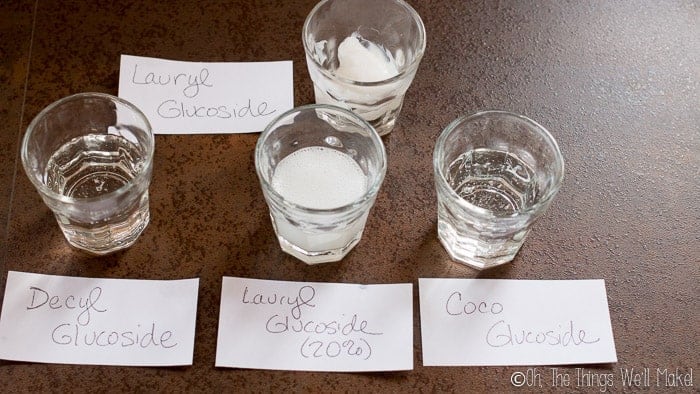
Disodium Laureth Sulfosuccinate
Disodium Laureth Sulfosuccinate is a gentle anionic surfactant that can be used in natural products (ECOCERT). It is a great alternative to SLS for a milder, more natural shampoo (or body wash, etc.). It has larger molecules than some of the other surfactants (like SLS) making it unable to penetrate and irritate the skin in the same way. It cleans and provides foam in products made for people with sensitive skin.
Coco Betaine
Coco betaine is a coconut based amphoteric surfactant. It’s mild and can help boost foam and increase the viscosity of products made with it. It’s very mild and provides for gentle cleansing. It’s completely biodegradable and has a pH around 6-8. It is also ECOCERT compatible so it can be used in the elaboration of “natural” and “organic” type products.
Sodium Coco Sulfate
Sodium coco sulfate is an anionic surfactant that is ECOCERT and BDIH friendly. It has a pH of 10-11 and is derived from coconut oil. It is a water-soluble surfactant that is sold in solid form. It’s usually used in non-soap shampoo bars and/or bar cleaners (syndet bars).
Plantapon® SF
Plantapon SF is a mix of vegetable-based surfactants (coconut, corn, and palm based) that can be used in a variety of gentle cleansing products like shampoos, shower gels, and facial cleansers. It includes sodium cocoamphoacetate, lauryl glucoside, sodium cocoyl glutamate, sodium lauryl glucose carboxylate, and glycerin. It has a pH between 6.5 and 7.5.
Because this is a mix of surfactants, it can be a good choice for those who are just delving into working with surfactants. You can easily mix up formulations without needing to buy a lot of raw materials or doing a lot of work. (I’ll work on getting up some recipes that use this as soon as I can.)

Completely natural surfactants
While not as effective as the other more processed surfactants derived from natural sources, those looking for a completely natural alternative may be interested in studying some of these natural surfactants. These plant based cleansers all have natural saponins that are a type of non-ionic surfactant. They can be used alone or combined with the other surfactants for a more effective final product.
Soap Nuts (Soap Berries, Aritha)
The fruits taken from the sapindus trees/shrubs from the lychee family have saponins which are natural non-ionic surfactants. They are usually called either soap nuts or soap berries, and they clean without creating much foam.
You can either throw a cloth bag of them in with your laundry to naturally wash your clothes, or you can steep them in warm water to extract a liquid that can be used for cleaning. Make just enough for what you’ll need and you can freeze the rest.
Liquid Yucca Extract
Liquid yucca extract is a natural non-ionic surfactant that comes from the yucca plant, a desert plant that has natural saponins of its own. While you can add it to your homemade shampoos, yucca extract is also used in gardening to help get nutrients to the roots of other plants by washing away concentrated salts that build up.
Shikakai powder
Shikakai powder is another plant with natural saponins which are natural non-ionic surfactants. It is normally used in hair care as a very natural “shampoo.” It naturally has a low pH which makes it ideal for hair care. It’s said to be good for all hair types, especially those that are prone to breakage and damage. Like with the other natural surfactants, you can either combine it with other surfactants or use it on its own. To use it on its own, you make a paste by mixing the powder with warm water and running it through your wet hair once it the paste has cooled. You then leave it to act for 10-15 minutes before rinsing it out. It may slightly darken hair.
Soapwort
Soapwort is another plant that has been used for many years as a soap alternative. It can be used to clean the skin, wash your hair, or even as a laundry soap. It’s especially good for delicate fabrics. To use soapwort, you need to make an infusion of the soapwort in water, and then you can use the resulting liquid as a liquid soap alternative.
 Español
Español



 How to Make Cold Brew Coffee (Without Special Equipment)
How to Make Cold Brew Coffee (Without Special Equipment)
HC
Thanks for the info on these natural surfactants. I have been looking for these quite a long period. Very much appreciated .
Tracy Ariza
You’re very welcome!
Adele de Swardt
Hi Tracy
Thank you for this very informative AND understandable article about surfactants (no i have to look for an article on natural preservatives 🙂 )
Q: i see soap nut liquid can be used as “shampoo”. What would you recommend – use only liquid as is OR would i have to add more ingredients? And how long would the mixture last stored in the shower?
Thanx, love your site!!!
Adele
Tracy Ariza
Hi Adele,
Thank you so much.
I’m working hard to make it more complete and organized, little by little.
You’re actually in luck! I just published my guide to natural surfactants a few minutes ago! ?
I’ve been working hard on it for a while now.
I think if you were going to try to use soap nut liquid as a shampoo, you would just use it as is. That’s the way I tried using it years ago. (It’s been awhile since I’ve been since working on other options.)
I can’t remember how well it went for me. I hadn’t been using shampoo anyway so I think it worked relatively well and better than just water alone. (Yes, I also tried that for awhile!)
Adele de Swardt
Ah thank you!! Found the article on preservatives 🙂 will have a read..
I might try soap nut shampoo but not no-poo (haha, not that brave)!
Thank you for your info, only site i found so far that does not just “copy and paste” other sites’ info. Grateful for your research!!
Chris
I’ve used soapnuts liquid (boiled soapnuts) as a shampoo and found that it’s far to harsh for my hair which is baby fine. Even in very very small quantities.
I’ve discovered that using any type of “real” shampoo on my hair removes all natural oils and tends to leave my hair flat and lifeless. Too bad it took 40 yrs to figure this out. lol
With regard to no poo, this works best for me, but not the baking soda variety. That will cause your hair to break and fallout.
Safe No Poo Shampoo
I steep 1 tbs dried rosemary, 1/2 tsp sage, and about an 1/8 tsp of soapwort in about 1/2 cup distilled or filtered water. Let it cool, strain, then add 1 tbs light rye flour and mix well. Massage into hair and let it sit for about 5 minutes before rinsing very very well.
My baby fine hair is now full, bouncy, and manageable. I will never go back to “real” shampoo again.
Shipra Agarwal
I am from India. Our ancients have been using Amla shikakai and reetha for washing hair since ages. I washed my hair made of these products as a child and has such beautiful hair. I believe I lost my hair after using chemical shampoos. Google and u will get receive how to make this shampoo of Amla reetha and shikakai. In India these products are very cheap
Shipra Agarwal
Damn auto corrects. Many typo errors due to it. Kindly ignore
marta
Hello Tracy,
I am looking into doing Syndet shampoo bars and I was a bit hopeless until I read this post! Shampoo bars (soap based) are quite harsh on my hair and I do not see them as a long term alternative. However I do like the idea to make a completely organic shampoo bar that is not soap-based. My understanding with this post is that this is still achievable if I choose to use a natural and plant derived ECOCERT surfactant (or more than one). Am I correct? In this case, to produce a solid shampoo bar would I need to use thickeners as cetyl alcohol? Or which ones you would recommend? Also, the other part that worries me in solid shampoo is the preservation system – do you have any advice on natural preservatives that could work effectively in a shampoo bar or would you go for a synthetic alternative?
Many thanks for sharing all your knowledge!
Tracy Ariza
Hi Marta,
To make a shampoo bar, you normally have to use solid surfactants. I have a recipe for a homemade shampoo bar up on the blog. It uses SCI and SLSA, which are both quite mild and safe.
I was actually taking a break from writing a post on natural preservatives, etc.- when I stopped to answer you here.
I use the same preservatives for the bar as I do with my lotions and liquid products. As the pH of a good shampoo syndet bar is in the perfect pH range for most preservatives, you can generally use whatever you like.
Astrid Fabre
Hello Tracy! Thank you very much for the information! I love your blog.
I was trying to figure out which surfactants to use for a multi-prupose cleaner and a glass cleaner. Thank you for all the help you might be able to give me it is greatly appreciated!
Astrid
Tracy Ariza, DDS
H Astrid,
You could probably use any of the mild surfactants, but in a very small amount to keep from streaking. I like using alcohol to help dry and avoid streaking too.
Serion
Hi Tracy, I’m on my way to make my very first soap and shampoo bar. Your page really is a great help for me. I wonder if I can make shampoo bar with only one surfactant (SCI noodle)? If yes, what is the maximum percentage for it? Thank you so much for your info. Warm hug from Indonesia!
Tracy Ariza
Hi Serion,
I would imagine that it would be fine. Perhaps I’ll try it myself next time to see what happens.
You’d probably have to keep the percentage of surfactants in the same range to keep the bar solid. While SCI is a great cleanser and does provide good lather, it’s quite mild so I don’t think it would be problematic to do it that way. It may change it slightly, but I don’t know for sure by how much as I haven’t tried it personally (yet)! 😉
Ishita
Hi Tracy, Yours is the 1st post I ever saw on surfactants and that also so informative. Thanks a ton for sharing such good information.
Now coming to my question, I am trying to make hot process soap and have tried adding coco betaine, sodium lactate,sodium citrate to my soaps to add lather to them.
My results have been varied-coco betaine did not help with lather at all, sodium citrate and sodium lactate made it a very drying soap.
Is there some surfactant which can help add lather and still be moisturising?
Tracy Ariza
Hi Ishita,
Thank you!
That’s a very interesting question!
Normally, the more natural surfactants provide less lather than soap which is an anionic surfactant. So, it makes sense that they wouldn’t improve lather.
I’ve actually never tried combining actual soap with other surfactants before and have really no idea about what would happen.
I do know that some of the surfactants don’t work as well at the high pH of soap.
So, perhaps, that’s another issue you could have.
So, you are adding the other surfactants after the saponification process has finished?
I actually think you’d be better off trying to adapt the actual soap recipe itself to make more lather if that’s what you are seeking by using oils like coconut oil that provide a lot of lather.
The solid surfactants I used in my homemade shampoo bar also provide a lot of lather, all while being gentle. Perhaps you can try one of those.
Denise Berger
wow! Finally a post on this very confusing subject that even I can understand! You hooked me. I’m looking pretty hard to find a recipe to make my own laundry detergent. There are tons out there using borax, washing soda and I’ve tried them but they’re too watery and they don’t clean well. I bought some coco glucoside and am now going to search your site to see what I find. Loved this article!
Tracy Ariza
Hi Denise,
Thank you so much. I’m happy you found it helpful!
I’m doing my best to keep studying and learn more and share more information. 😉
I hope it goes well for you!
Emma Rieth
Very informative! Would the properties of some of the natural surfactants (yucca in particular) survive saponification to use in a soap or shampoo bar?
Tracy Ariza
Hi Emma,
That’s a very interesting question. I don’t really know for sure.
The problem is, because soap already lathers, it would be really difficult to know for sure, even if you were to try it.
The lathering and cleansing abilities of soap are already stronger than those of something like yucca so it wouldn’t make much of a difference, even if those properties would survive.
I have seen the natural surfactants like yucca combined with some of the other milder surfactants in some products. (Although I suspect they do it mostly for marketing appeal.)
j chandler
tracy,
i hope you and your husband and family are doing very well!
i haven’t talked to you since last year and i’m amazed at the improvements you’ve made in your site!
i love your info on glucosides, they are my favorite new best friend! i have found many versions of the polyglucosides or alkyl glucosides! just found hexyl glucoside, capryl glucoside, and your three—-decyl, lauryl, and coco! many recipes and commercial brands use your 3—– decyl, lauryl, and coco plus capryl glucosides in their formula from bath and body, laundry, and dish products! i’m definitely switching from my favorites in dish and laundry commercial products to homemade because of ewg’s rating and the fact that they cause cancer with formaldehyde and ethylene oxide and dioxin in them!
as i told you before, i’ve had cancer 3 times so this is a definite concern to me! i’ve switched my toothpaste, my bath and body, deodorant, etc… already to homemade and all have been amazing to my improved health! the next project is getting the cheapest glucoside with the best performance in cleaning without any irritation! i can’t use any of your bath and body recipes though, not because they are not excellent, but because at least one ingredient irritates my skin! i used decyl alone in my body, bath, and shampoo recipes plus other non-detergent additives! hand soap has decyl plus sodium cocoyl isethionate and i love it! the dish and laundry will need a stronger blend—probably the hexyl and capryl, but, hexyl is 81 dollars per gallon as opposed to 30 to 40 dollars per the other glucosides! also, i decided to try the zinc ricinoleate in my deodorant formula and i like it, but, it’s definitely more waxy than i like and under performs compared to zinc oxide deodorant! continued good luck in your ventures in and through life! i’m approaching 64 and life is an amazing blessing which i try not to take for granted and take advantage of every precious moment i can while GOD lets me live! hope to continually write to you and converse through this site!
Tracy Ariza
Hello!
It’s great to hear from you!
I really like coco glucoside a lot. It’s quite gentle.
Yes, I tend to add other ingredients to my recipes- not because they are necessary, but to show people the sorts of things you can add. I do try to explain my choices, though, to help people adapt the recipes as needed to better suit them. So hopefully you’ll still be able to get what you need from my posts. 😉
As for the deo recipe- that’s interesting to me. You’re the first person who has told me that they think the zinc oxide is more effective than the ricinoleate.
I had one couple tell me that they make it with only the ZR because they are sensitive to the ZO, and they liked it much better that way. (Which, in itself is unusual, I think, as ZO is pretty mild and used in diaper creams!)
On the other hand, others have told me that they have made it both with and without the ZR, and it works better for them with the ZR.
I’m tempted to try a batch with only ZR out of curiosity! I’ve always made it as written just because it works for me and I don’t want to waste ingredients. On the other hand, I’m curious. 😉
My experimentation could be used to help others, though.
What I think is obvious, though, is that we are all different and react differently to different ingredients- it’s finding the best ones for us that’s important.
For hand washing dishes, and for body, I often use my homemade liquid soap. That’s another option for when you don’t want to use the detergents. It may be more cleansing than the mild detergents as it is an anionic surfactant.
I really wish you the best and many healthy years to come! 🙂
Anne
Thank you so much for taking the time to share your knowledge with us all Tracy. I can’t belief that anyone would get impatient with you.
Anne. London
Tracy Ariza
Hi Anne,
Thank you! You’re too kind. 🙂
I try my best.