Trying to avoid SLS and other harsh surfactants in your cosmetics? There are many mild, natural surfactants available. Learn about the different types of natural surfactants, with a list of my favorites.

What is a surfactant?
There are many types of surfactants and they are used for many different purposes, but they all share one quality: they help increase the wetting properties of a liquid. Surfactants can be found almost everywhere. You can find them in everything from detergents and shampoos to toothpaste and even conditioners.
Surfactant definition:
(A surfactant,) also called surface-active agent, (is a ) substance such as a detergent that, when added to a liquid, reduces its surface tension, thereby increasing its spreading and wetting properties. (Encyclopaedia Britannica)
Surfactant function
Some surfactants are emulsifiers, others are foaming agents (and some may actually do the opposite of those functions). Some act as detergents, while others act as insecticides or fungicides. Some help with solubilizing (small amounts of oils into water, for example) and others help increase viscosity.
How do surfactants work?
Surfactants affect the surface tension of liquids to increase wetting.
Why would you want to increase wetting?
Normally, when you spray water on a surface like a window, rather than spread evenly over the surface, the water will bead up. That’s because of the surface tension of the water. The molecules of the water come together in a stable configuration and are attracted to each other. When you are trying to clean that window, though, that beading isn’t helping you. You want the water to spread evenly over the surface to better clean it. You also want something that can grab onto the grease and dirt on whatever surface you are trying to clean.
Micelles in surfactants
Surfactants affect the surface tension that is making the water bead up rather than spread out. They have a water-loving head and an fat (oil) loving tail. They come together in structures called micelles.

I already explained a bit about how the micelles in surfactants work in my micellar water recipe, but for those who haven’t read that post, let me give you a quick, simplified explanation. The water-loving heads of the micelles bond with the water while the oil-loving tails on the inside of the micelles bond with the grease and grime. That pulls the grease and grime into the center of the micelles out of contact with the water, making them easier to rinse away.
You’ll also find that hot water helps clean better because the hot water helps melt the fats which makes it easier for them to be brought into the micelles.
Types of surfactants
There are four main types of surfactants, each behaving somewhat differently, and some with completely different functions. The detergent-like surfactants tend to be the anionic, non-ionic and amphoteric surfactants. Some cationic surfactants are used as emulsifiers and are great for hair conditioners. (I use BTMS, a cationic surfactant, in my hair conditioner recipe.)
These are classified based on the charge of the polar head of the surfactant which can have a positive charge (cationic), a negative charge (anionic), or no charge (non-inonic). Amphoteric surfactants have both a cationic and anionic part attached to the same molecule.
- Anionic – Anionic surfactants are the most commonly used surfactants because they tend to provide the best cleaning power and the most foam. You’ve probably heard people talking about one of the most commonly used anionic surfactants, SLS (Sodium lauryl sulfate or Sodium Laureth Sulfate). It can be found in everything from shampoos and shower gels to even toothpaste. I’ve also shown you how to make soap (many types by now!), another anionic surfactant.
Anionic surfactants can be harsher on the skin, which is why they are often combined with other types of milder surfactants. - Nonionic – The second most commonly used surfactants are nonionic surfactants. They don’t ionize in water or aqueous solutions. Nonionic surfactants are gentler when cleaning. Because they don’t carry a charge, they are the most compatible with other types of surfactants. Recently, sugar-based nonionic surfactants have been developed to offer a safer, non-toxic alternative to some of the more harsh surfactants on the market up until now.
- Cationic – Cationic surfactants don’t generally give foaming like the other types of surfactants. They are often used in hair care products (mainly conditioners and anti-static products because they don’t provide the foaming for use in shampoos) because their positive charge is attracted to the negative charge in hair. This makes it difficult to completely wash them from your hair, so some stays behind to help reduce friction between hairs which, in turn, reduces the amount of electrostatic charge in hair. This helps make hair more manageable and helps prevent damage.
Cationic surfactants aren’t usually compatible with anionic surfactants! - Amphoteric – Amphoteric surfactants can carry either a positive or negative charge depending on the pH of your product. Despite that, they are still compatible with all of the other types of surfactants. These tend to be very mild surfactants which is why they are usually combined with other surfactants. While amphoteric surfactants may not give a lot of foam on their own, they can help boost the foam of the other surfactants. Amphoteric surfactants are often combined with anionic surfactants to reduce their harshness and help stabilize their foam.

Natural surfactants list:
Natural surfactants can be derived from many types of plants. Common sources are coconut or palm, but they can also be derived from other types of fruits and vegetables.
There are many natural surfactants on the market today, and with increased consumer demand, I imagine that many more will be available in time. I have tried many of them, but today I’ll focus on some of my favorites. I like these surfactants because they are gentle, they tend to be easier to find, and they work well together. You can use these in everything from gentle shampoos to shower gels, facial cleanser, and baby washes.
Choosing your surfactants
Keep in mind that many of these surfactants are not palm free, so you’ll want to source them from places that allow for sustainable methods of obtaining their materials. I buy surfactants that have been certified sustainable by RSPO (Roundtable on Sustainable Palm Oil) standards.
Another thing to keep in mind is that these surfactants can differ from manufacturer to manufacturer. The names are polymeric and aren’t referring to an exact structure. Some places will use different plants as the origin of elaborating each surfactant, and the way each surfactant cleans, solubilizes, etc. can vary depending on where you buy it from. I’ll be describing these surfactants based on my suppliers, but you’ll want to check on the specifications of the surfactant you are buying if it’s important to you to know what plants have been used to derive them, the pH, the concentration, etc. Use this list as a general guideline!
Along those lines, while mine are listed as ECOCERT approved, that may also be dependent upon the manufacturer of each surfactant.
I’ll be updating this list and adding more surfactants as I use them and learn more about them. For now, though, this should give you a good starting point to understanding what we are going to be working with.
Coco Glucoside
Coco Glucoside is a non-ionic surfactant that is obtained from coconut oil and fruit sugars, but it can also be obtained from either potato or corn. It is a very gentle, foamy cleanser and is completely biodegradable. You can use it in products that you want to have an ECOCERT certification. It has an alkaline pH (around 12) which makes it self-preserving as is, but you will probably have to adjust the final pH of products using it to pull it into a range more suitable for your skin or hair (and you’ll need to add a preservative).
Decyl Glucoside
Decyl Glucoside is very similar to coco glucoside (non-ionic and ECOCERT compatible), but it has a shorter chain length. It creates less foam (its foam is less stable) than coco glucoside but it does add more viscosity to a product. It is derived from coconut oil and glucose and is completely biodegradable. It can be used in all sorts of shampoos, gels, baby products, etc.
Lauryl Glucoside
Lauryl Glucoside is very similar to the other 2 glucosides I’ve mentioned. It has a longer chain length and more viscosity. It takes longer to foam than the other two, but it also has the most stable foam. While it is also a mild cleanser, it isn’t as mild as the other 2 alkyl polyglucosides.
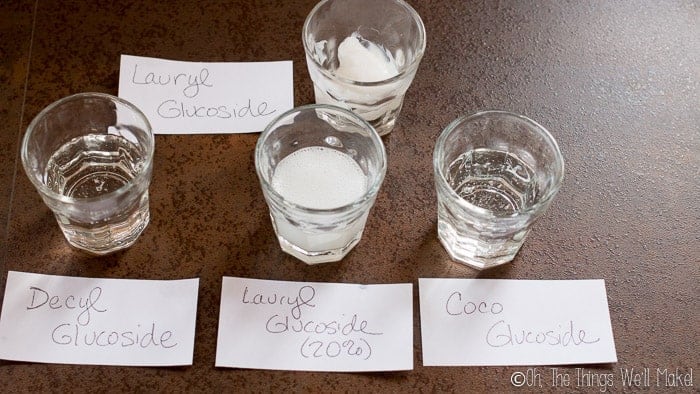
Disodium Laureth Sulfosuccinate
Disodium Laureth Sulfosuccinate is a gentle anionic surfactant that can be used in natural products (ECOCERT). It is a great alternative to SLS for a milder, more natural shampoo (or body wash, etc.). It has larger molecules than some of the other surfactants (like SLS) making it unable to penetrate and irritate the skin in the same way. It cleans and provides foam in products made for people with sensitive skin.
Coco Betaine
Coco betaine is a coconut based amphoteric surfactant. It’s mild and can help boost foam and increase the viscosity of products made with it. It’s very mild and provides for gentle cleansing. It’s completely biodegradable and has a pH around 6-8. It is also ECOCERT compatible so it can be used in the elaboration of “natural” and “organic” type products.
Sodium Coco Sulfate
Sodium coco sulfate is an anionic surfactant that is ECOCERT and BDIH friendly. It has a pH of 10-11 and is derived from coconut oil. It is a water-soluble surfactant that is sold in solid form. It’s usually used in non-soap shampoo bars and/or bar cleaners (syndet bars).
Plantapon® SF
Plantapon SF is a mix of vegetable-based surfactants (coconut, corn, and palm based) that can be used in a variety of gentle cleansing products like shampoos, shower gels, and facial cleansers. It includes sodium cocoamphoacetate, lauryl glucoside, sodium cocoyl glutamate, sodium lauryl glucose carboxylate, and glycerin. It has a pH between 6.5 and 7.5.
Because this is a mix of surfactants, it can be a good choice for those who are just delving into working with surfactants. You can easily mix up formulations without needing to buy a lot of raw materials or doing a lot of work. (I’ll work on getting up some recipes that use this as soon as I can.)

Completely natural surfactants
While not as effective as the other more processed surfactants derived from natural sources, those looking for a completely natural alternative may be interested in studying some of these natural surfactants. These plant based cleansers all have natural saponins that are a type of non-ionic surfactant. They can be used alone or combined with the other surfactants for a more effective final product.
Soap Nuts (Soap Berries, Aritha)
The fruits taken from the sapindus trees/shrubs from the lychee family have saponins which are natural non-ionic surfactants. They are usually called either soap nuts or soap berries, and they clean without creating much foam.
You can either throw a cloth bag of them in with your laundry to naturally wash your clothes, or you can steep them in warm water to extract a liquid that can be used for cleaning. Make just enough for what you’ll need and you can freeze the rest.
Liquid Yucca Extract
Liquid yucca extract is a natural non-ionic surfactant that comes from the yucca plant, a desert plant that has natural saponins of its own. While you can add it to your homemade shampoos, yucca extract is also used in gardening to help get nutrients to the roots of other plants by washing away concentrated salts that build up.
Shikakai powder
Shikakai powder is another plant with natural saponins which are natural non-ionic surfactants. It is normally used in hair care as a very natural “shampoo.” It naturally has a low pH which makes it ideal for hair care. It’s said to be good for all hair types, especially those that are prone to breakage and damage. Like with the other natural surfactants, you can either combine it with other surfactants or use it on its own. To use it on its own, you make a paste by mixing the powder with warm water and running it through your wet hair once it the paste has cooled. You then leave it to act for 10-15 minutes before rinsing it out. It may slightly darken hair.
Soapwort
Soapwort is another plant that has been used for many years as a soap alternative. It can be used to clean the skin, wash your hair, or even as a laundry soap. It’s especially good for delicate fabrics. To use soapwort, you need to make an infusion of the soapwort in water, and then you can use the resulting liquid as a liquid soap alternative.
 Español
Español



 How to Make Cold Brew Coffee (Without Special Equipment)
How to Make Cold Brew Coffee (Without Special Equipment)
Desi
Hello can I make a gentle jelly cleanser with coco bentaine ,peg 7 glyceryl cocoate,and cocoglucoside?
Tracy Ariza, DDS
Hi Desi,
When you say “Jelly cleanser,” I’m not sure what, exactly, you are trying to make. Is it just a regular gel cleanser or something with a different texture? Could you link to a similar product so I can see?
Jodie
Hello, thank you for your research. Your website has been helpful! I am a trichologist creating a hair care line. First rule of thumb is do no harm. I am looking for surfactants that will not leave a residue, not block follicle, no citrus, not affect the color and create a little foam or lather. Just a very pure cleanse that will not disrupt natural ph. I’d like to keep the ph low since water is generally more alkaline than the hair. If I use non-ionic surfactants, will they be less likely to bond to harsh hard water elements like aluminum, calcium, lead, copper, etc since minerals are “positively charged”? What combo would you recommend for what I am trying to achieve?
Tracy Ariza, DDS
Ho Jodie,
I usually use a mix of an amphoteric and a non-ionic. (In some cases, for a more thorough cleanse, I have added an anionic, but I use it more for my shampoo bars.)
I think they work well as one can help stabilize the foam of the other. And, yes, you’re correct that non-ionics can be a bit more resistant to hard water issues.
You can play with amounts and combinations, though.
Jordan M
Hello,
In regards to the natural surfactants, would adding a preservative to the shampoo mix prolong the shelf life?
Tracy Ariza, DDS
Hi Jordan,
Any product that has water in it- or that can be easily contaminated with water, really needs a preservative to be safely used.
I have a post that may help you with more information about when, why, and how to use a preservative in your products.
Kal
I have made a shampoo with just Cocamidopropyl betaine as the only cleanser, a made up recipe on my own, I’ve used it a few times and it seems to be working fine… however I’ve never heard of anyone using just one single surfactant in a shampoo, so is this safe to use? Or will it not work well long term?
Tracy Ariza, DDS
Hi Kal,
Yes, it’s fine to do that.
The reason they are normally combined is that some add more lather, others may help stabilize that lather, some can make the harsher ones gentler, etc.
In the end, though, you can use whatever works for you. I tend to combine 2 or 3 different types in the hopes of getting more balance, but because these surfactants are generally mild, if they’re doing the job, then go with it!
Navyashree
Hello,
I would like to know if I can mix soapnut, shikkai and other surfactant like coco betaine and make a mild shampoo.
Tracy Ariza, DDS
Yes, you could experiment with those. Be careful when using shikakai as it can be very irritating to eyes.
Jamie van Buren
Thank you for this post! I was recently diagnosed with an allergy to coconut derivatives and fragrance and I have not been unable to find one shampoo on the market that excludes these ingredients, especially coconut derivatives. I’ve been researching options and trying to figure out if i could make my own shampoo.
I’m wondering if I could use Disodium Laureth Sulfosuccinate and/or Disodium Lauroyl Glutamate for this purpose. Do you have any recommendations? Thank you so much!
Tracy Ariza, DDS
Hi Jamie,
You’d have to ask the supplier. I think even the same surfactant can be derived from slightly different ingredients from supplier to supplier. I haven’t been able to find a good answer about which non- coconut-based surfactants are available.
Jamie
Hello,
I was recently diagnosed with allergies – including an allergy to coconut derivatives. There are a total of 0 shampoos I have found that are safe for me. I am trying to create my own. Are there any surfactants that are not derived from coconut that could help create a lathering type of soap/shampoo? Thank you!
Tracy Ariza, DDS
Hi Jamie,
I’ve asked this in my cosmetic formulation classes, and, unfortunately, I wasn’t able to get an answer for any to recommend.
Even the same surfactants can be derived from different ingredients, depending on the supplier. Most (if not all) do have either coconut or palm or both, though.
You’d have to ask suppliers directly if they had something available, I’m afraid. I wish I had a better answer for you!
Luci Flint
Using a surfactant base with coconut oil to mix foaming bars or bath bars does that cause problems over time in drains and pipes? I know coconut oil in solid form when using for oil pulling is not to be spit down the sink. I want to make soaps with lavender on our on my property but do not want to use lye. I want to control what I put in my bath bars. Looking for an organic solution
Tracy Ariza, DDS
Hi Luci,
When you make soap, the coconut oil is combined with the lye and forms a chemical reaction, so you no longer have neither coconut oil nor lye in the final product. So, no, it’s not a problem to use coconut oil soap in your sink. The same goes for coconut based surfactants. With time, yes, some surfactants can build up over time, especially if you have hard water, but it’s not the same process as with coconut oil.
Unfortunately, it’s impossible to make a true soap without lye. (I have more information about that in my post about lye.)
If you want to use natural detergent based surfactants, you can use some of the others mentioned in this post.
James
Hi Tracy, Been following up your blog a lot. You have inspired me to make my own soap. I need to ask you regarding this surfactants, I am trying to thicken my castille soap into a gel form. Will using the surfactants instead of water to dillute the soap paste be a good idea or there is a better way to tranform soap paste into a gel form? Thank you.
Tracy Ariza, DDS
Hi James,
I don’t’ normally combine other surfactants with soap as they tend to work best at different pH ranges. In any case, most surfactants are quite liquid too and need to be thickened with either a gum or with salt.
I haven’t done much experimentation with thickening soap, but some people are successful using a salt solution. Others thicken with a gum like xanthan or guar gum.
Chynita
Hello, this article has been extremely informative so Thank You for taking the time out to do this. What do you recommend to made a wash that is safe for babies decyl glycoside or coco glycoside?
Tracy Ariza, DDS
Hi Chynita,
Either of those should be fine. They are very similar and both quite mild.
I have a baby wash recipe up on the blog, and I’ve made it with both. I don’t really notice much of a difference either way.